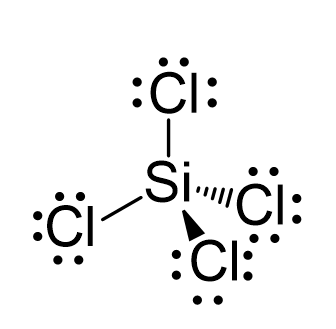
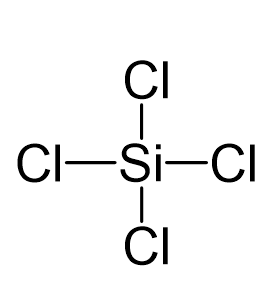
Silicon tetrachloride SiCl4: Molecular Geometry - Hybridization - Molecular Weight - Molecular Formula - Bond Pairs - Lone Pairs - Lewis structure –
We all know that CCl4 and SiCl4 both compound remains in Tetrahedral structure , with a hybridization of sp³ in the central atom. Now , Si is a much larger atom than
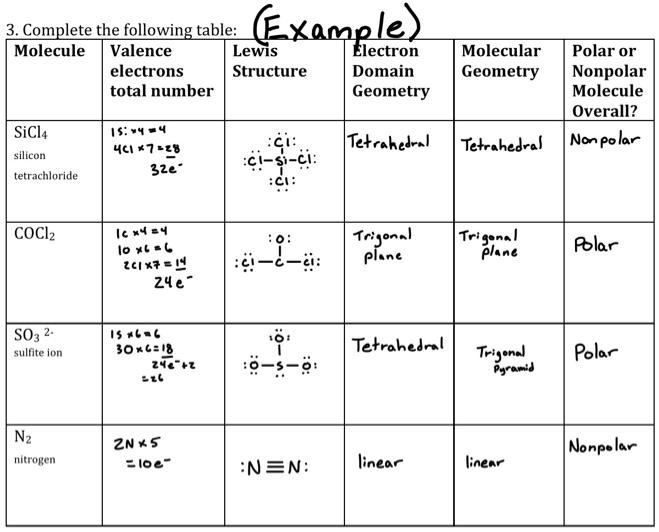
SOLVED: Complete the following table: Molecule Valence Lewis Electrons Structure Domain Total Number Geometry Molecular Geometry Polar or Nonpolar Molecule Overall? Nonpolar SiCl4 Silicon Tetrachloride 4 7-2-4 3-2-4 Tetrahedral Tetrahedral Nonpolar COCl2
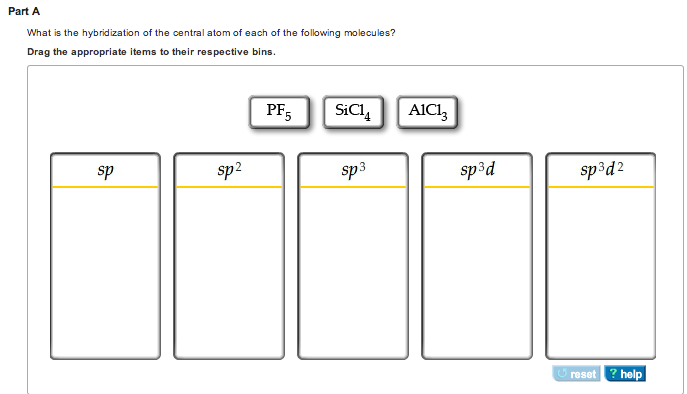
What is the hybridization of the central atom of each of the following molecules? - Home Work Help - Learn CBSE Forum
Q. Which of the following pair has same hybridisation. 1] BF3 NF3 2] SF4 SiCl4 3] ClO4 ClO2 4] CO2 SiO2
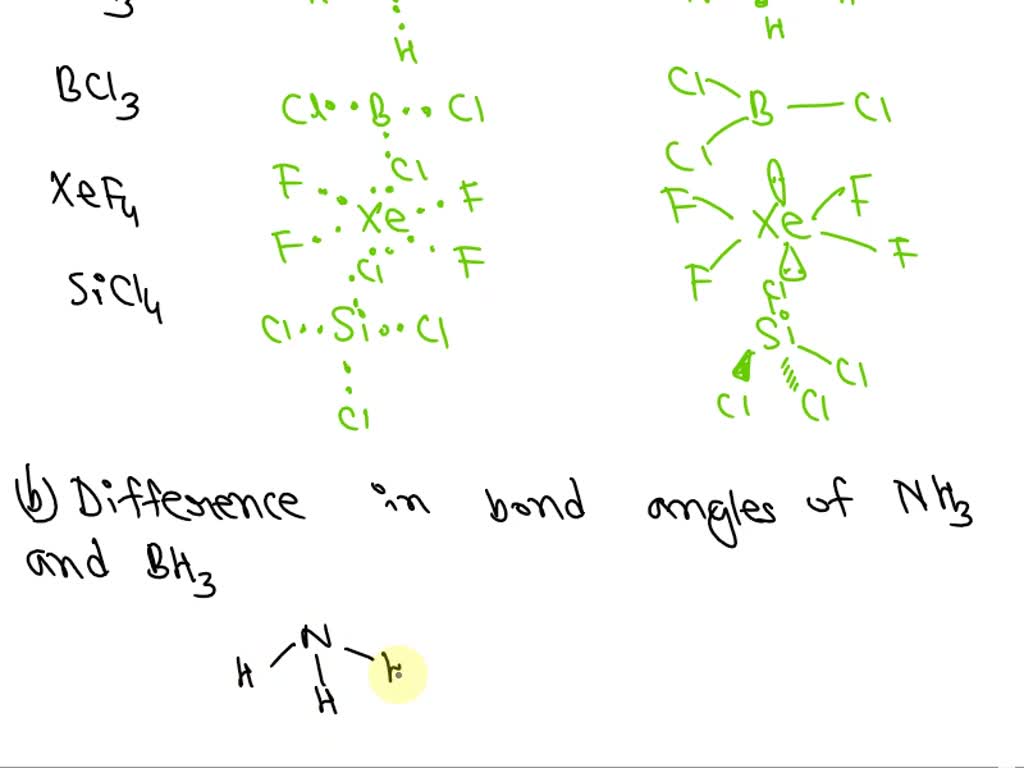
SOLVED: a) Draw the Lewis structures and molecular shapes of NH3, BH3, PCl5, XeF4 and SiCl4, and indicate the hybridization types and geometric shapes. b) Compare the bond angles in NH3 and

SOLVED: Hybridization of Si in SiCl4 is sp3d2, sp3d2, sp3d2, sp3d2, sp3d2, sp3d2, sp3d2, sp3d2, sp3d2, sp3d2 and in: SiCl2 is sp3d2

SOLVED: 4a) Using Valence Bond Theory, show the hybridization and bonding scheme for silicon tetrachloride (SiCl4): (a) write the atomic orbital diagram for the central atom, (b) circle the atomic orbitals that