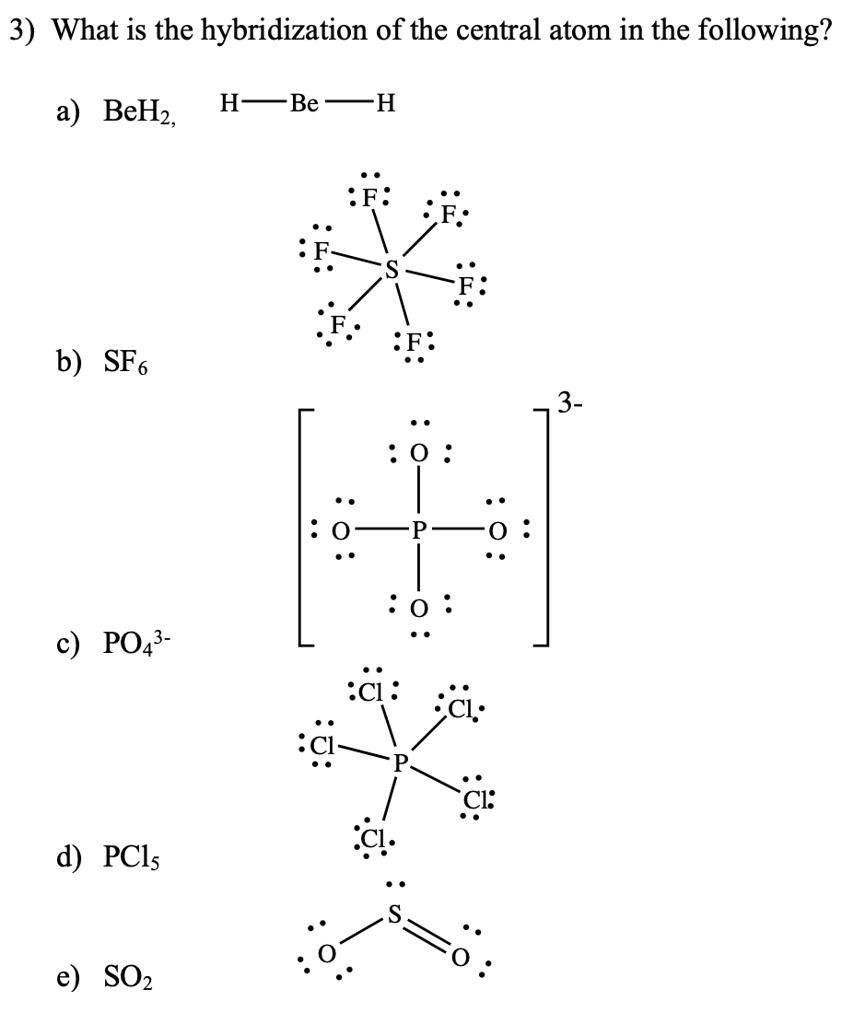
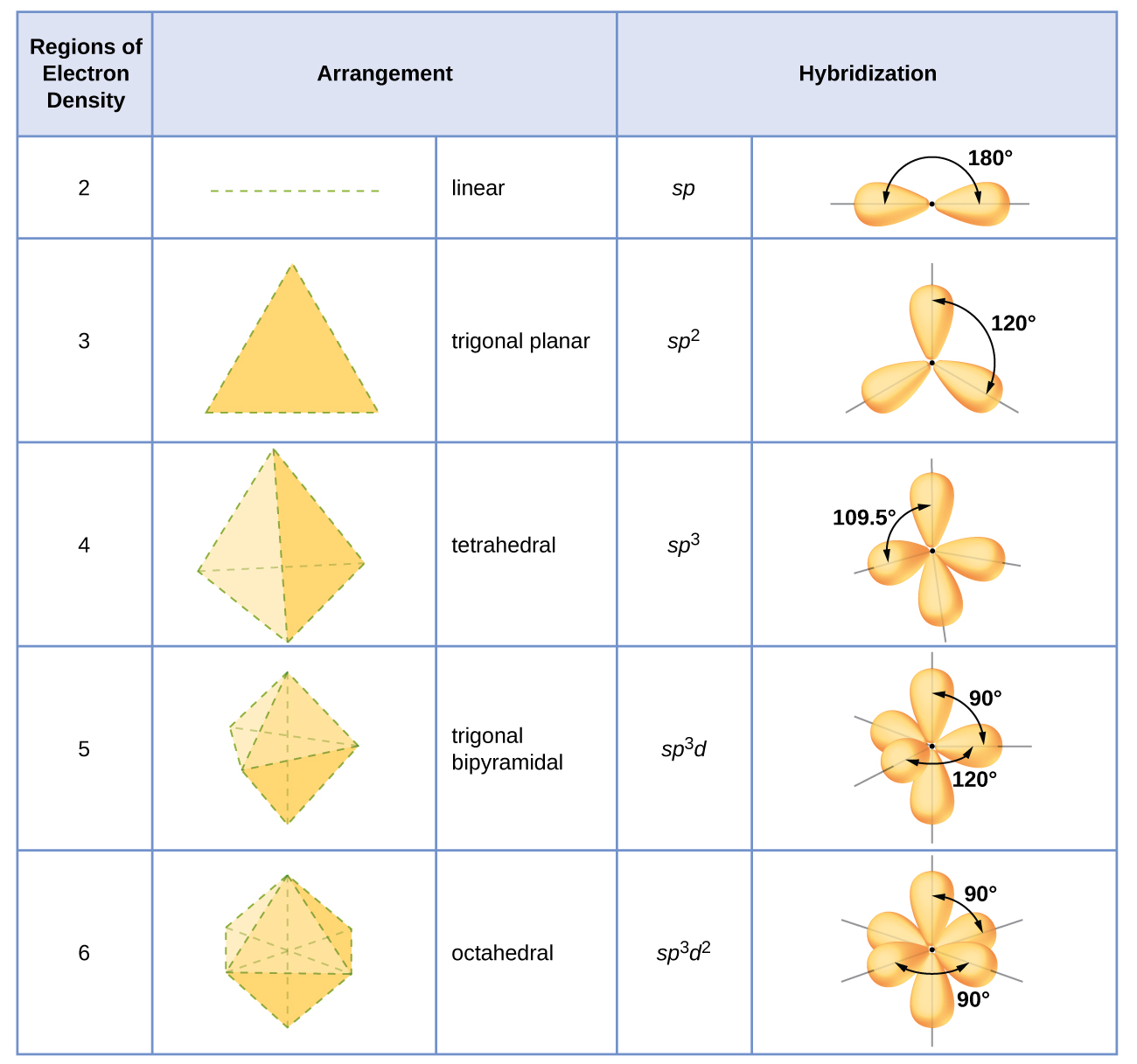
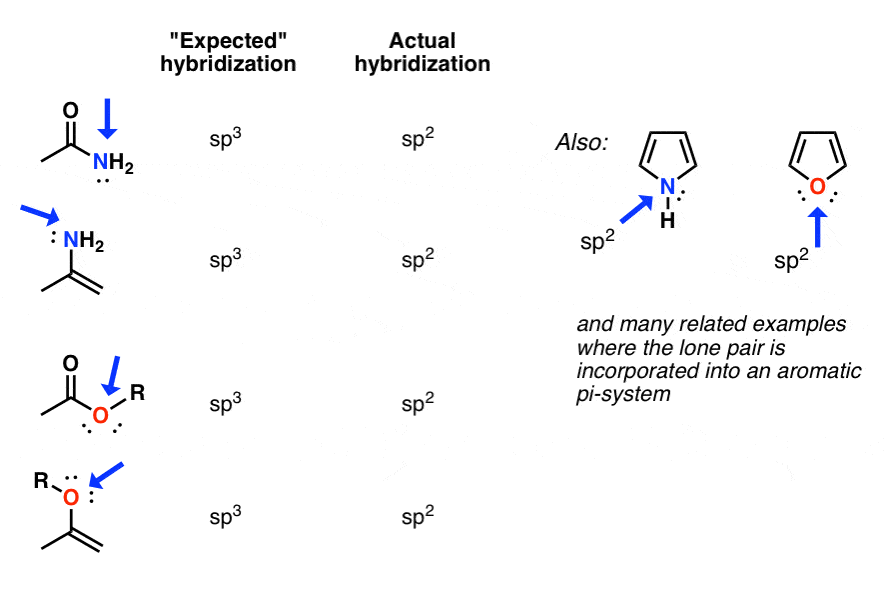
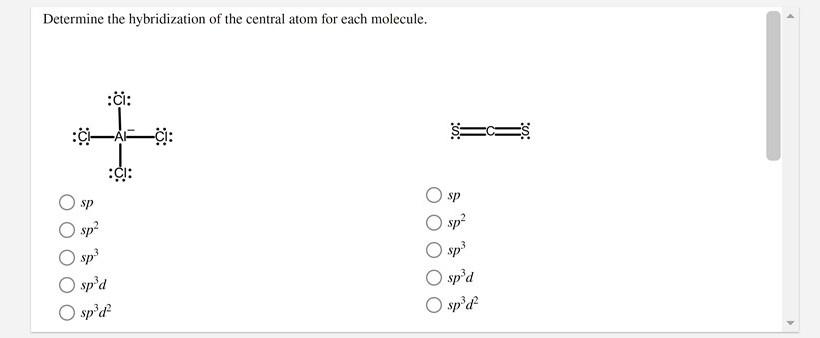
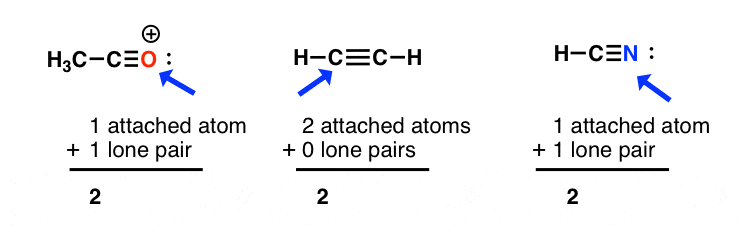
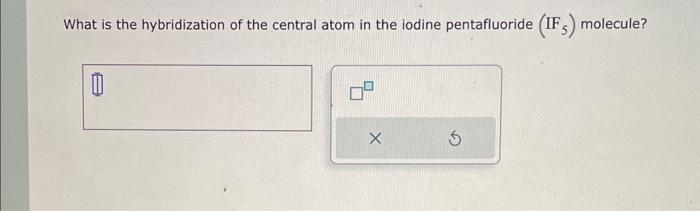
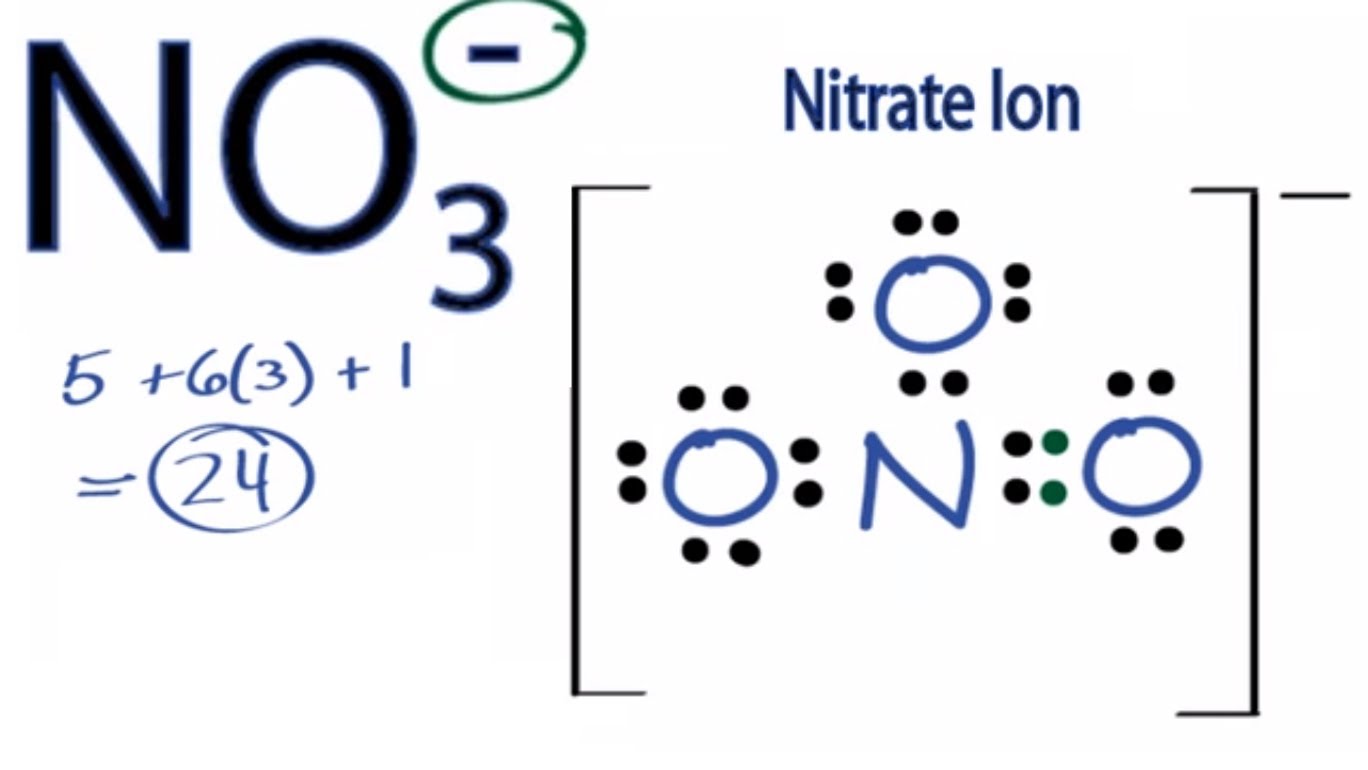
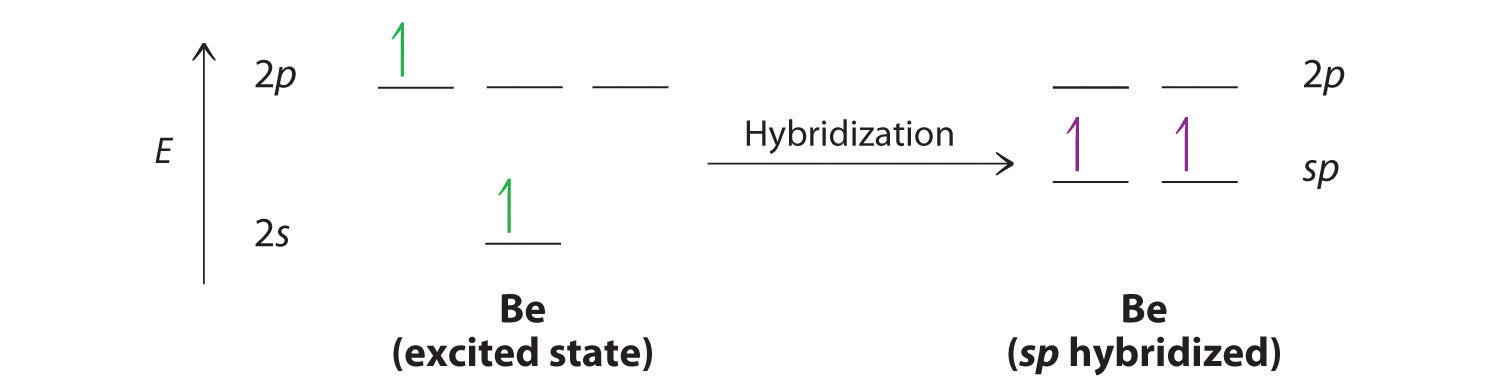
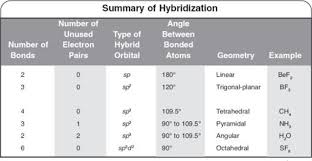
In which of the following pairs of molecules/ions, the central atoms have sp^{2} hybridization?BF_{3} and NO{_{2}}^{-}NO{_{2}}^{-} and NH_{3}BF_{3} and NH{_{2}}^{-}NH{_{2}}^{-} and H_{2}O
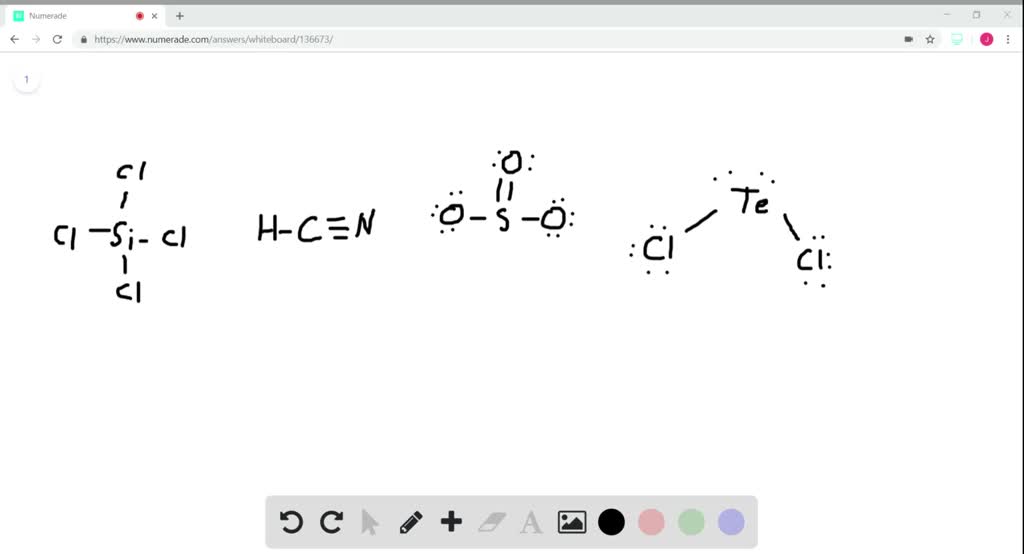
19. In which one of the following species the central atom has the type of hybridization which is not the same as that present in other three? 1)SF4 2)I3 3)SbCl3 4)PCl5