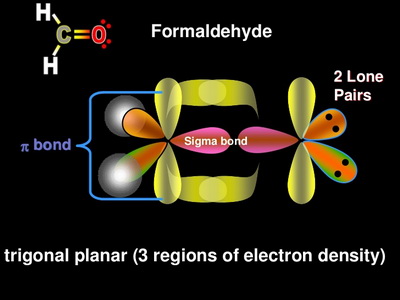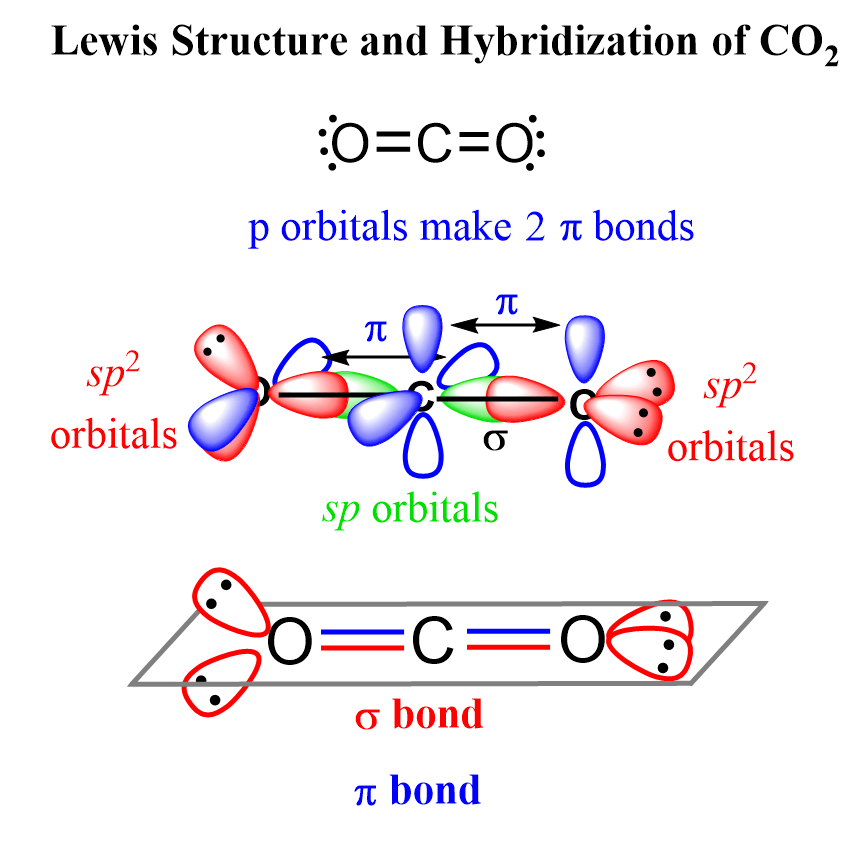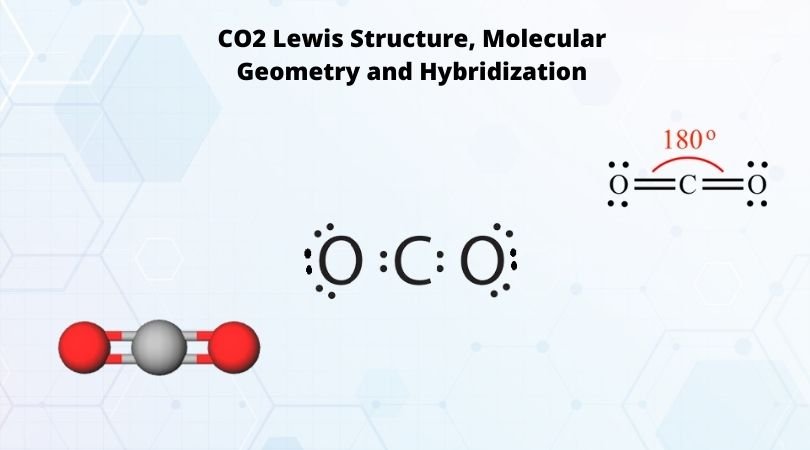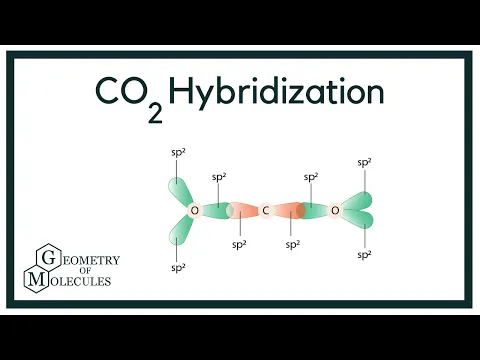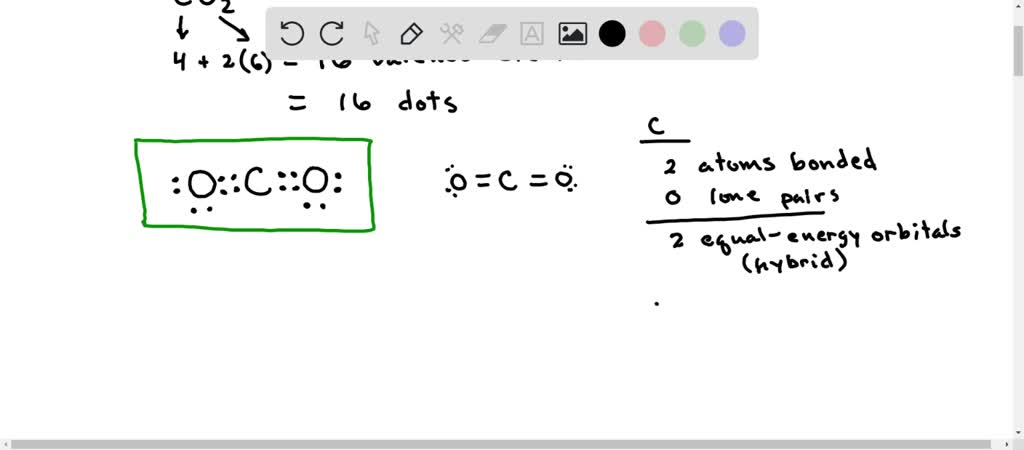
SOLVED: Draw a Lewis structure for CO2. Determine the hybridization on the carbon atom. Which orbitals of the carbon atom remain remain unhybridized? How many sigma and pi bonds are formed?
In which of the following molecules does the carbon atom have sp hybridization: HCN, CH4, CO2, and CH2O? - Quora

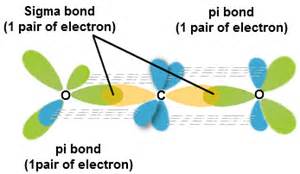

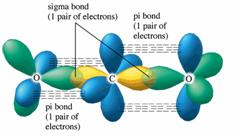

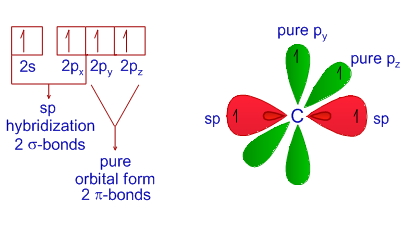




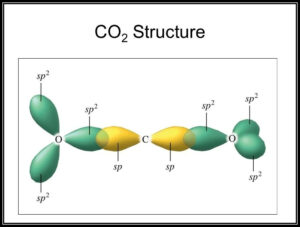
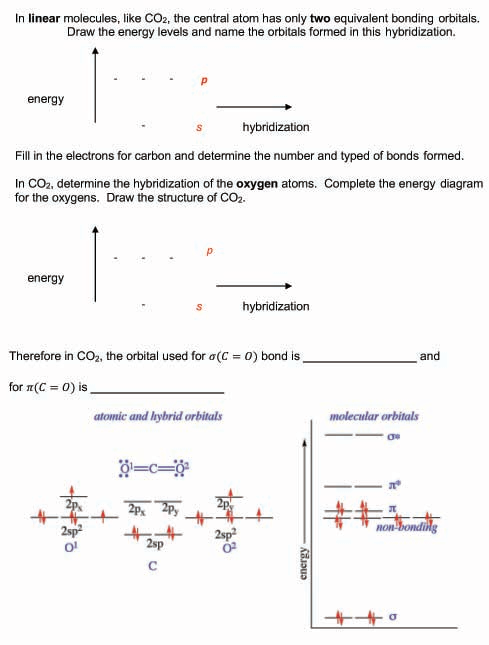

![SOLVED] In CO2 hybridization of carbon is sp sp2 sp3 None of t - Self Study 365 SOLVED] In CO2 hybridization of carbon is sp sp2 sp3 None of t - Self Study 365](https://static.tllms.com/moodle-migration/47309_7fc775895c5b6da80cbe1e11e0d869d746d07138_S-1.PNG)